- Home/
- SSC & Railways/
- Article
Name the Strongest Acid in the World
By BYJU'S Exam Prep
Updated on: September 25th, 2023
Fuloroantimonic is the strongest acid in the world.
Acid
- Acids are a type of chemical that can act as a proton donor or acceptor.
- Strong acids range in pH from 1 to 3.
- The most potent acid to be found is fluoroantimonic acid. The primary constituents are hydrogen, fluorine, and antimony.
- It has a variety of various cations and anions and is a combination of hydrogen fluoride and antimony pentafluoride.
- Its pH is 20 quintillion times higher than sulfuric acid with a purity of 100%.
- This acid serves as a catalyst for the synthesis of synthetic materials, fuels, and biology.
- The weak connection between hydrogen and fluorine makes this acid especially harmful and corrosive.
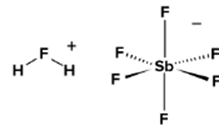
Hence, Fuloroantimonic is the strongest acid in the world.
Acid – Any hydrogen-containing chemical that has the ability to donate a proton (hydrogen ion) to another chemical is considered an acid.
Table of content
Properties of Acids –
- Acids have a tendency to corrode.
- They are effective electrical conductors.
- Their pH levels are never greater than 7.
- These chemicals react with metals to form hydrogen gas.
- Acids have a sour flavour.
- Examples: Sulfuric acid [H2SO4], Hydrochloric acid [HCl], Acetic acid [CH3COOH].
Uses of Acids –
- Acetic acid is diluted into vinegar, which is used in many household processes. The main application for it is as a food preservative.
- Lemon juice and orange juice both include citric acid as a primary ingredient. Additionally, it can be used to preserve food.
- Acid sulphate is frequently used in batteries. This acid is frequently found in the batteries used to start car engines.
- Sulfuric acid and nitric acid are used in the industrial manufacturing of fertilisers, paints, dyes, and explosives.
- A significant component of many soft drinks is phosphoric acid.
Summary:-
Name the Strongest Acid in the World
Fuloroantimonic is the strongest acid in the world. Any chemical that contains hydrogen and is capable of donating a proton (hydrogen ion) to another chemical is referred to be an acid. Acids are used as food preservatives and in batteries to start car engines.
Related Questions:-
- A Copper Wire Has A Diameter Of 05mm And Resistivity Of 16 X 10 8 Omega M What Will Be The Length
- A Particle Is Moving In A Circular Path Of Radius R Its Displacement After Half A Circle Would Be
- A Ray Of Light Traveling In The Air Enters Obliquely Into Water Does The Light Ray Bend Towards
- Abc Is A Right Angled Triangle In Which A 900 And Ab Ac Find B And C
- A Hockey Ball Of Mass 200 G Travelling At 10 Ms 1 Is Struck By A Hockey Stick So As To Return
- Check Whether 6n Can End With The Digit 0 For Any Natural Number N
- Describe The Work Of A Farmer With 1 Hectare Of Land

