- Home/
- GATE MECHANICAL/
- GATE ME/
- Article
Heat Treatment Process of Steel: Types, Steps of Heating
By BYJU'S Exam Prep
Updated on: September 25th, 2023

The Heat Treatment process is a combination of heating and cooling operations applied to steel. To improve the Mechanical properties of steel, some alloying elements were used. However, steel properties can also be controlled without adding other elements. This is accomplished by the heat treatment process of steel, where controlled heating and cooling operations are done.
The history of the swordsmith and cutler crafts makes it abundantly evident that the precise process of hardening steel, by plunging the solid red-hot steel into water (producing martensite’), and toughening steel by tempering the quench-hardened steel at a moderate temperature, have been experimentally known and utilized for thousands of years. The art of heat treatment process of steel must have been known precisely to people in many regions of the world several hundreds of years ago because the steel objects made by them, such as daggers, knives, swords, etc., could be favorably compared with the best that can be manufactured utilizing most recent procedures and equipment.
Download Formulas for GATE Mechanical Engineering-Engineering Mechanics
Table of content
What is Heat Treatment Process?
Heat treatment of metals and alloys is done to them enhance their machinability, reduce internal stresses, or improve their mechanical properties. Heat treatment of steel can also alter the properties of carbon steel significantly.
Heat treatment of steel is an effort to maximize its efficiency under harsh service conditions. Steel is an excellent versatile engineering material used in many products. The fact that steel’s characteristics may be controlled and modified at will (within reason, reasonable limits) by heat treatment accounts for a significant portion of steel’s versatility.
Download Formulas for GATE Mechanical Engineering – Machine Design
Steps in Heat Treatment Process
The heat treatment process consists of three basic steps. All of these heat-treating procedures are similar in that they all involve the heating and cooling of steel. The methods, however, differ in terms of the heating temperatures used, the cooling speeds employed, and the results.
1. Heating Steel to a Predetermined Temperature: Most heat treatments begin with heating the steel to a predetermined temperature, usually the austenitizing temperature, which depends on:
- The composition of the steel
- The heat treatment is applied to the steel, such as annealing, normalizing, hardening, etc.
The steel’s desired qualities will determine the type of heat treatment used.
2. Soaking: It holds the steel at the austenitizing temperature for a specific minimum without grain development. Soaking time is the shortest time required for the entire section (including the center) of the component to achieve the desired microstructure, i.e., the pre-existing phases transition to fine-grained and homogeneous austenite without grain development.
3. Cooling at a Predetermined Rate: The cooling rate fixed for a part depends on:
- Heat treatment for the alloy that is fixed based on the properties that the steel will eventually acquire,
- Steel composition and
- The thickness of the part.
Different Types of Heat Treatment Process
Various heat treatment processes are used to enhance the properties of steel depending upon the temperature, time of heating, cooling and nature of the steps used. The five basic processes of heat-treatment of steel are given below:
- Annealing
- Normalizing
- Hardening
- Tempering
- Case Hardening
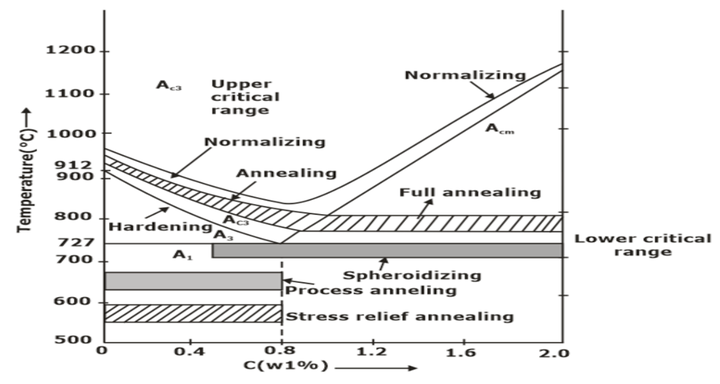
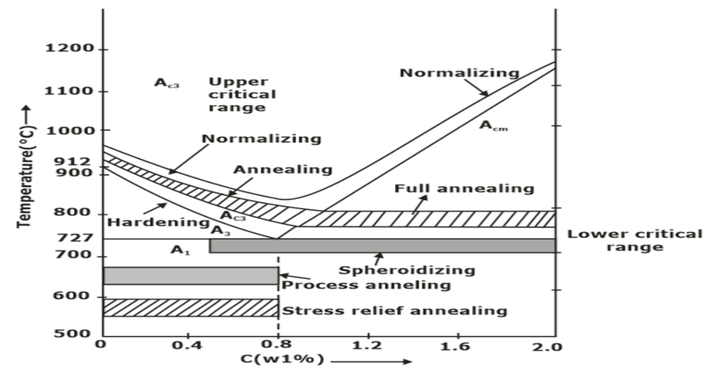
Annealing
Annealing is a heat treatment of steel used to soften the material. Along with softening, any internal stresses will be removed.
Its carbon content will determine the approximate temperatures at which the steel sample should be heated.
Soaking time should be set at 3-4 minutes per mm thickness of the material’s cross-section. The workpiece is permitted to cool inside the furnace only after the electrical power or oil supply is turned off. As a result, the workpiece cools down relatively slowly. Due to grain development, this process softens the material and increases its ductility.
Annealing is a heat treatment process generally used to:
- Relieve internal stresses
- Increase softness, ductility and toughness
- Produce a specific microstructure.
It consists of three stages:
- Stage I- Heating to the desired temperature
- Stage II- Holding or Soaking’ at that temperature
- Stage III- Cooling usually to room temperature.
It is employed for low and medium carbon steels experiencing plastic deformation during forming operations.
(a) Full Annealing
Hypoeutectoid steel and hypereutectoid steel are heated above 50°C of upper and lower critical lines, respectively. Then, the component is cooled slowly in the furnace. Hence coarse grains are obtained, resulting in decreased strength and hardness.
(b) Process Annealing
The hypo eutectoid steel is heated below the lower critical line. It relieves the back stresses, and no change in microstructure occurs. It is only for low-carbon steel.
(c) Spherodise Annealing
It improves the machinability of medium and high carbon steel. The specimen is heated just below the lower critical temperature. The carbide in steel is transformed into a globular form.
(d) Diffusion Annealing
It makes the chemical composition uniform. Next, the sample is heated to 1150°C, kept at that temperature for 6-8 hours, and cooled slowly in the furnace.
Normalizing
Normalizing is a type of heat treatment of steel that requires heating to the same temperatures as indicated for annealing (except for high carbon steel specimens, which must be heated to much higher temperatures than recommended for annealing, especially as the carbon percentage in the sample increases), soaking, and finally cooling in still air. The primary goals of normalizing are grain refinement and eliminating internal stresses.
- Steels that have plastically deformed, e.g., a rolling operation, consist of grains of pearlite and a pro eutectoid phase, which are irregularly shaped and relatively large and vary in size.
- Normalizing is used to refine these grains (i.e. to decrease average grain size) and produce a more uniform and desirable size distribution.
- The microstructure formed is fine-grained pearlite with a pro eutectoid Phase.
- It is accomplished by heating alloy (55°C to 85°C) above the upper critical temperature; after sufficient time has been given to alloy to transform to austenite completely, the treatment is terminated by cooling in air. It raises the hardness and strength of alloy but decreases the ductility of steel.
Hardening
Hardening is a steel heat treatment accomplished through heating (to the same temperatures as annealing) and soaking. The workpiece is removed from the furnace and rapidly cooled in a tank of cold water or oil while being forcefully agitated in the water/oil. (This process of chilling is known as quenching). The workpiece hardens as a result. However, the work piece’s carbon content needs at least 0.25 per cent to be hardened. Therefore, this method cannot be used to harden dead mild steel.
Additionally, mild steel will become significantly more challenging for specimens with more than 0.25 per cent carbon. The resultant hardness will be higher than the carbon proportion. Pieces that have been hardened become highly brittle, a significant drawback. They frequently fail when in use.
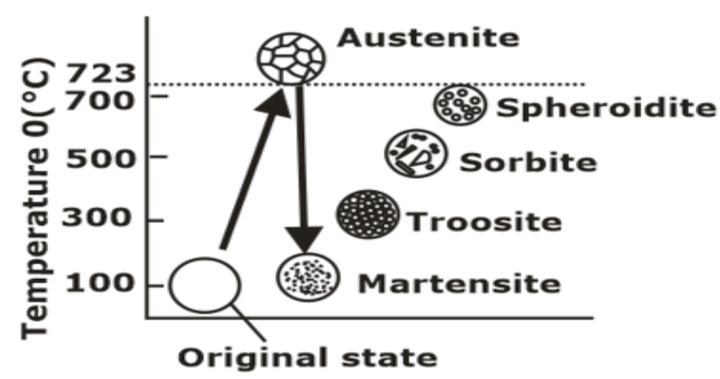
Tempering
Tempering is a type of heat treatment of steel that means giving up some hardness while shedding a significant quantity of brittleness gained during the hardening process. Hardness and brittleness are traded off for the hardened component to perform well over time. Tempering involves heating the carbon steel part to temperatures ranging from 150° to 600° C (depending on how much trade-off is necessary) and cooling the component in an oil or salt bath or even in the air.
It is done:
- To relieve residual stress
- To improve flexibility
- Toughness is increased.
(a) High-temperature tempering (500-700°C)
The resulting structure is Sorbite; residual stress completely vanished.
Tempering of martensite.
(b) Medium temperature tempering (300-500°C)>
- Resulting in Troostite formation.
- After tempering, work is cooled to water, leading to enhanced endurance.
- Due to the increase in endurance limit, thus used for making spring steels and die steels.
(c) Low-temperature tempering (150°C-300°C):
- Holding time is 1-3 hrs.
- Apart from achieved properties in tempering, this process provides additional wear resistance”.
- It enhances true tensile strength and hardness change to a small extent.
- It is used for measuring cutting tools.
Case hardening or Surface Hardening
Case hardening is a steel heat treatment that hardens only those carbon steels with a carbon content of 0.25% or more. The workpiece is packed in charcoal and heated similarly to annealing in this technique. For a few hours, it is maintained at such a high temperature. As a result, depending on how long the workpiece is heated, carbon penetrates the surface to a depth of one or two mm.
- Its main objective is to achieve hard surfaces with a relatively soft core, and a hard surface is called a case.
- Hard surface provides good wear resistance, while good toughness is the result of softcore. There are various treatments to impart surface hardness which are discussed below:
(a) Carburizing
- It is applied to low carbon steels having up to 0.18% C.
- The temperature range is between 870-950°C and results in the formation of the Austenite phase, which has maximum solubility of carbon.
(i). Pack carburizing:
- Here, the pieces to be hardened are surrounded by a carburizing mixture and then packed in a steel box.
- It is heated to around 870-950°C.
- Carburizing mixture: 50% charcoal + 20% BaCO3 + 5% CaCO3 + 5-12% Na2CO3
Pack carburizing
(ii). Gas Carburizing
- Here work part is treated in an atmosphere of carbon and hydrocarbon gases such as CH4, butane etc.
- Here, the workpiece is heated to a temperature of 950°C and held for 3-12 hours.
CH4 → 2H2 + C (atomic)
(iii). Liquid Carburizing
- Here, the part is heated to 950°C and kept in a molten salt bath with 20% NaCN, which gives Carbon (C) & Nitrogen (N).
- Carbon and a small amount of Nitrogen diffuse into the surface at this temperature.
- In carburizing operations, carbon can be penetrated up to 0.3 mm with hardness obtained around (55-65 HRC).
(b)Nitriding
- It is done by heating steel in the atmosphere of NH3 gas.
- Pieces to be nitrided are placed in an airtight container. Workpieces are heated to a temperature in the range (of 500-600°C), at which NH3 dissociates into 2NH3 → 2N + 3H2
- This atomic nitrogen diffuses to the surface, making the case hardened.
(c) Cyaniding
- Here work part is immersed in a molten salt bath containing sodium cyanide (NaCN), which is heated to 820-860°C.
- This is usually followed by water quenching.
- The cyanide both consists of: 20-30% NaCN + 25-50% NaCl + 25-50% Na2CO3
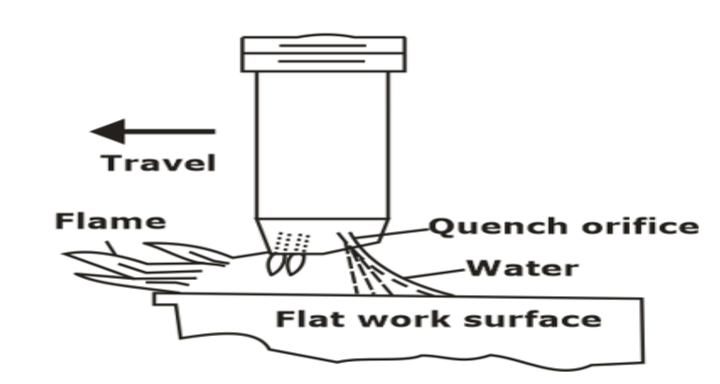
(d) Flame or Induction hardening
- Workpieces are heated in an induction furnace surrounded by copper coils which are water cooled. High-frequency AC is passed through Cu coils and thus alternate magnetic field set up which induce eddy currents on the surfaces.
- Heat is generated on the surface by eddy currents.
- The surface of the workpiece is heated in the austenitic range and then quenched immediately to form martensite.
- The structure of the core remains unchanged because it is not affected by heat.
- The heating rate is 300°/sec for hypo eutectoid steel; subsequently, the heating time is minimal (2-50 sec.). Quenching temperature depends upon the rate of heating. If the rate is 500°C/sec, it is 1000°C; if it is 250°C, then 900°C.
- The process consists of heating the surface of high carbon steel with a high-temperature gas flame at 2400-3500°C, followed by immediate cooling in air or water.
- An oxy-acetylene torch supplies heat. The heat is supplied so quickly to the surface that the core remains unaffected.
- The thickness of the hardened layer is 2-4 mm, and its structure is the martensite.