Heat transfer takes place as per (a) Zeroth law of thermodynamics, (b) First law of thermodynamics, (c) Second law of thermodynamics, (d) Kirchhoff’s law
By BYJU'S Exam Prep
Updated on: October 17th, 2023
Heat transfer takes place as per the Second law of thermodynamics. As Heat transfers from Higher Temperature to Lower Temperature
Zeroth law of thermodynamics:
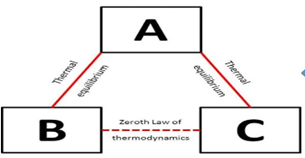
According to the zeroth law of thermodynamics, two thermodynamic systems are in thermal equilibrium with one another if they are both in thermal equilibrium with a third system. The measurement of temperature is based on this law.
Thermodynamics’ first law:
According to the basic law of thermodynamics, energy can only be transferred between different forms and cannot be generated or destroyed in a closed system.
The law of conservation of energy is restated in the first law of thermodynamics, which goes as follows:
ΔQ = ΔW + ΔU
Second law of thermodynamics:
The Second Law of Thermodynamics states that absent some external aid, heat always moves in one direction (from a body at a higher temperature to one at a lower temperature).
Entropy is a concept that is used to quantify the direction of flow and is stated as
Δ S = ∫dQ/T
Δ Q = heat exchange
Δ W = work done due to expansion
ΔU = internal energy of the system
ΔS = change in entropy
T = temperature
This is the conclusive point for the given statements
Zeroth law – Concept of temperature
First law – Concept of internal energy
Second law – Concept of entropy/ direction of heat flow
Summary:
Heat transfer takes place as per (a) Zeroth law of thermodynamics, (b) First law of thermodynamics, (c) Second law of thermodynamics, (d) Kirchhoff’s law
According to the Second Law of Thermodynamics, heat transmission occurs. Second Law of Thermodynamics States that Heat Transfers from Higher Temperature to Lower Temperature.


