Ozone Layer; Ozone Hole Recovery
The ozone layer is a region of the stratosphere. The ozone layer helps in absorbing the Ultraviolet (UV) rays of the sun which are harmful to human beings. UV rays can cause sunburn and damage the DNA, the ozone layer absorbs most of the harmful radiations and protects us. The layer prohibits at least 98% of harmful Ultraviolet radiations, coming from the sun to earth and protects humans, animals along with the plants from these radiations.
What is the Ozone hole?
Ozone molecules in the atmosphere are produced over the tropics and then spread enveloping the earth’s atmosphere. But due to prevailing stratospheric winds, it is transferred towards the poles. When the concentration of ozone is reduced at a particular place, it is considered an Ozone hole. In other words, one can say that the damaged area of the Ozone layer is known as the Ozone hole. Its concentration is measured in Dobson units. The largest ozone hole is above Antarctica.
To understand the ozone layer presence, let us have a glance at the atmospheric layers:
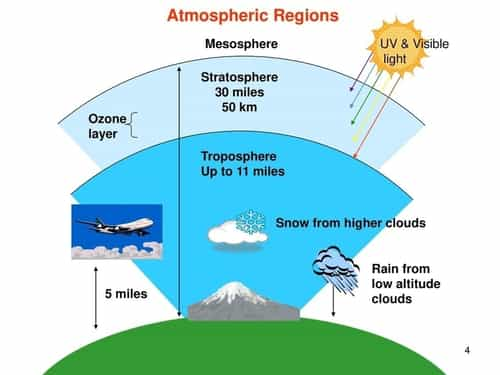
Ozone layer’s thickness varies geographically, it is thinner at the equator and thicker near the poles. Wherever, the layer thins, the harmful radiations – UV rays pass through it and reach the earth’s surface. Based on the wavelength absorbed, the UV radiations are of three types:
- UV-C (280-100nm) is entirely screened out by a combination of dioxygen and ozone).
- UV-B (315-280nm) majorly screened out by ozone. But the part of it passes which helps form Vitamin D.
- UV-A (400-315 nm) is transparent to all. It reaches the earth’s surface completely.
The UV radiation is also harmful not only to humans but also to plants and animals.

How is the Ozone (O3) formed?
Ozone is produced naturally in the stratosphere when highly energetic solar radiation strikes molecules of oxygen, O2, and cause the two oxygen atoms to split apart in a process called photolysis. Ozone (O3) is made up of three layers of oxygen. The layer formed by the Ozone is known as the Ozone layer. If a freed atom collides with another O2, it joins up, forming ozone O3. The concentration of the ozone layer is nearly 10 ppm and in the Earth’s atmosphere, it is present at about 0.3 ppm.
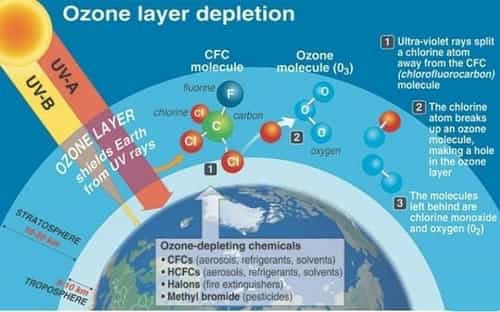
Ozone is produced naturally in the stratosphere when highly energetic solar radiation strikes molecules of oxygen, O2, and causes the two oxygen atoms to split apart in a process called photolysis. If a freed atom collides with another O2, it joins up, forming ozone O3. The concentration of the ozone layer is nearly 10 ppm and in the Earth’s atmosphere, it is present in about 0.3 ppm.
How does the ozone layer deplete?
The gradual thinning, reduction, or wearing out of the Ozone layer in the stratosphere due to various human activities is called Ozone depletion. The depletion of the ozone layer is caused mainly by Green House Gases (GHGs) like CO2 and methane, other GHGs that includes CFCs that are used in aerosols such as household sprays, hair sprays, cans, fridges and making plastics etc. The chemical reaction caused by the CFC’s and halons break down ozone molecules and thus, results in the reducing capacity of UV rays of the Ozone layer. Other gases that contribute to the depletion of the ozone layer are Nitric oxide (NO), Nitrous Oxide (N2O), Hydroxyl (OH), Atomic Chlorine (Cl) from Chlorofluorocarbons (CFCs)and Atomic Bromine (Br) from Bromofluorocarbons etc.
- The thinning or reduction of Ozone depletion is most prominent in the Polar Regions, especially over Antarctica.
- The severe depletion of the Ozone layer in any particular area is called Ozone Hole.
Generally, we find thinning or depletion of the ozone layer to the extent of ozone hole creation over the southern pole i.e. above Antarctica. This is due to the chemical reactions that take place primarily on the surface of polar stratospheric clouds, ice particles, or liquid droplets that form at high altitudes in extreme cold. But in 2019, an unusually stable polar vortex in the northern polar region due to low temperature resulted in similar chemical reactions involving bromine and chlorine atoms (generally from CFCs and other gases) resulted in ozone depletion leading to the ozone hole over the Arctic at the northern pole.
Major causes of Ozone Depletion
- The main reason is the release of chlorofluorocarbons (CFCs) and halon gases in the atmosphere formerly found in aerosol spray cans and refrigerants.
- Excessive use of Nitrogenous Fertilizers.
- Use of Supersonic aircraft.
- Nuclear explosions.
Effects of Ozone Depletion
- It will magnify the effects of Ultraviolet Rays on human health.
- It may cause sunburn, skin cancer or cataracts.
- Excessive Ultraviolet rays may damage our eyes.
- It may cause global warming.
- It can destroy plants.
Measures to protect Ozone layer depletion
- Reducing the use of Ozone Depletion Substances (ODS).
- Effective implementation of Montreal Protocol concluded in 1987 for protection of the Ozone layer.
- Use of eco-friendly chemicals in the refrigeration and air-conditioning industry.
- Revamping the industries producing the chlorofluorocarbons (CFCs).
- Use of eco-friendly fuel in the automobile industry.
Efforts to protect Ozone layer depletion
By the early 1980s, scientists discovered that the Ozone layer was thinning over Antarctica. In 1987, countries around the world came together to fix the problem through the Montreal protocol. The international treaty banned the production of ozone-depleting chemicals i.e. Chlorofluorocarbons. These chemicals had been in use for decades and they stay near the atmosphere for a long time. So, it took years to see the effects. In 2000, the ozone hole was the largest ever observed by scientists. But conditions slowly improved. Studies show that the Ozone hole should recover in the middle of the 21st century.
Some of the important international programmes
- Vienna Conference
- It took place in Vienna, Austria in 1985.
- The ozone hole was first discovered during this time.
- It was signed for the protection of the ozone layer.
- It does not include legally binding reduction goals.
- Saving the Ozone layer Conference
- This happened in London, 1985.
- It dealt with the withdrawal of all the ozone-depleting substances.
- Helsinki Conference
- It took place in May 1989.
- To revise the Montreal Protocol, the Helsinki Conference happened.
- There was an agreement to eliminate CFCs
- Montreal Protocol
As discussed earlier, recent studies show that the ozone layer is recovering because of the Montreal Protocol, which was signed by 197 countries in 1987 to minimize the use of ozone-depleting substances. It was the first treaty in the United Nations to achieve universal ratification. The pact made has helped in pausing the depletion of the ozone layer.
Key highlights of the Montreal Protocol:
- It took place in 1987.
- It is a protocol to the Ozone Layer Vienna Convention.
- This was signed by 197 countries so as to minimize the use of ozone-depleting substances.
- Targets only ozone-depleting substances (Not GHG i.e. Hydro Fluorocarbons)
- All the developed countries decided to reduce the use of CFC’s. Regarded as the Universal treaty (Ratified by all UN countries)
- Montreal Protocol is considered as the first treaty in the history of the United Nations which achieved universal ratification.
- It has helped in pausing the depletion of the Ozone layer.
- Legally binding
Kigali Agreement
In this agreement, another set of substances named hydrofluorocarbons (HFCs), were introduced as non-ozone depleting alternatives. This is done to ensure the timely phase-out of CFCs and CFCs. HFCs are now widespread in air conditioners, refrigerators, aerosols, foams and other products. But these have high global warming potential. Uncontrolled growth in HFC emissions, therefore, challenges efforts to keep global temperature rise at or below 2°C this century. Urgent action on HFCs is needed to protect the climate system.
- It was signed in 2016 to Reduce Ozone Layer Depletion
- It amends the 1987 Montreal Protocol.
- Its objective is to decrease hydrofluorocarbons (HFCs) by approximately 80-85 per cent by the end of 2045.
- It is a legally binding agreement between the signatory parties with non-compliance measures.
- The Kigali Amendment to the Montreal Protocol is legally binding and came into force from January 1, 2019.
- India has ratified this agreement.
A recent study shows that the ozone layer is recovering as the world is under lockdown due to the ongoing viral outbreak and also because of the efforts are in place to curb the depletion. The researchers and scholars are of the opinion that the ozone layer is recovering due to the Montreal Protocol. The latest study mentions that countries must take most and more robust steps to reduce the emission of ozone-depleting substances in order to reduce Carbon footprint. There is no denying that lockdowns across multiple countries currently curb the ongoing viral outbreak has had a positive impact on the environment.
To boost the preparation of all our users, we have come up with some free video (Live Class) series.
Here are the links
MP राज्य परीक्षाओं के लिए करंट अफेयर्स
मध्य प्रदेश राज्य परीक्षाओं के लिए 2000 सबसे महत्वपूर्ण प्रश्न
More from us
Get Unlimited access to 45+ Mock Tests-BYJU'S Exam Prep Test Series




Comments
write a comment