- Home/
- CDS & Defence/
- Article
Write structures of different dihalogen derivatives of propane.
By BYJU'S Exam Prep
Updated on: September 25th, 2023
Propane has four distinct dihalogen derivatives. n-Propane, dimethyl methane, and propyl hydride are further names for propane. It functions as a food propellant and is a gas molecular entity. French chemist Marcellin Berthelot made the discovery of propane in 1857. n-Propane is a colorless gas with a little aroma of petroleum. It is dispersible in benzene, chloroform, and ethyl ether. It is typically produced as a by-product of two other operations, namely the processing of natural gas and the refining of petroleum. It is frequently utilised as fuel.
Table of content
Structures of Various Derivatives
Below is a diagram showing the structures of various derivatives.
(i) 1, 1-Dibromopropane
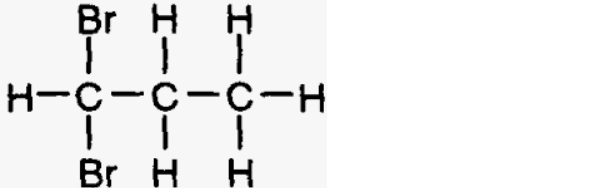
(ii) 2, 2-Dibromopropane
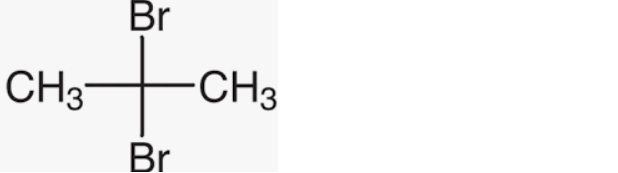
(iii) 1, 2-Dibromopropane

(iv) 1, 3-Dibromopropane
![]()
Uses of Propane
- Food additives use propane.
- utilised as a part of liquid petroleum gas.
- utilised in the production of ethylene and propylene.
- utilised as fuel for cutting and welding processes.
- used as the foundational building block for chemical synthesis.
- used as an energy source in water heaters and automobiles.
- utilised as homemade explosives.
- applied to lawn movers.
- the refrigerant used in.
- Applied to the campaign.
- used as a fuel for industry.
Health Hazards of Propane
Gangrene may result from the propane’s vaporising liquid. If the concentration in the air is higher than 10%, it may make you feel queasy, and a larger dose may make you asphyxiate. When heated, it causes containers to explode. If compared to air, its vapors are heavier. This substance is highly flammable.
Summary:
Write structures of different dihalogen derivatives of propane.
The structures of different dihalogen derivatives of propane are given above. Propane is a vaporising liquid that will make humans feel queasy and in heavy dosage will make them asphyxiate.
