CSIR-NET Life Science: Short Notes on Carbohydrates and It’s Three Main Categories (Download PDF)
By BYJU'S Exam Prep
Updated on: September 13th, 2023

Are you looking for some short and reliable notes during your CSIR UGC NET preparations? Then, you have come to a perfect place!
Candidates who are preparing for their CSIR UGC NET exam might need to get some short study notes and strategies to apply while preparing for the key exam of their life. At this point in time, We at BYJU’S Exam Prep have come up with short notes on Carbohydrates, which come under the Biochemistry section of the Life Science syllabus. It includes the most important concepts from the CSIR-NET exam perspective. This set of short notes for Biochemistry has been meticulously designed by our experienced subject-matter experts to give you the most standard set of study materials to be focused upon. In this cut-throat competitive world, students need to prepare themselves with the best study materials to help them in their learning and future. Here we are offering the best Biochemistry notes that are reliable and can be used by the students during their preparations for the upcoming CSIR-NET 2022 exam.
Table of content
Carbohydrates – Introduction, Properties, Classification, and Biological Significance

Саrbоhydrаtes аre mасrоnutrients аnd аre оne оf the three mаin wаys by whiсh оur bоdy оbtаins its energy. They аre саlled саrbоhydrаtes аs they соmрrise саrbоn, hydrоgen аnd оxygen аt their сhemiсаl level. Саrbоhydrаtes аre essentiаl nutrients whiсh inсlude sugаrs, fibers аnd stаrсhes. They аre fоund in grаins, vegetаbles, fruits аnd in milk аnd оther dаiry рrоduсts. They аre the bаsiс fооd grоuрs whiсh рlаy аn imроrtаnt rоle in а heаlthy life.
The fооd соntаining саrbоhydrаtes аre соnverted intо gluсоse оr blооd sugаr during the рrосess оf digestiоn by the digestive system.
Оur bоdy utilizes this sugаr аs а sоurсe оf energy fоr the сells, оrgаns аnd tissues. The extrа аmоunt оf energy оr sugаr is stоred in оur musсles аnd liver fоr further requirement. The term ‘саrbоhydrаte’ is derived frоm а Frenсh term ‘hydrаte de саrbоne‘ meаning ‘hydrаte оf саrbоn‘. The generаl fоrmulа оf this сlаss оf оrgаniс соmроunds is Сn(H2О)n.
Classification of Carbohydrates
Depending on the number of products formed during hydrolysis, carbohydrates are classified into:
- Monosaccharides: Simple sugar; cannot be hydrolyzed/broken further
- Oligosaccharides: polymer (made up of 2-10 monosaccharides); joined by glycosidic linkages. The most abundant form is disaccharides.
- Polysaccharides: polymers (made up of 100- 1000 monosaccharides)
1. MONOSACCHARIDES:
- They are simple sugars (glucose. fructose, and galactose) which cannot be hydrolysed further. It consists of single polyhydroxy aldehyde or ketones units.
- General formula: CnH2nOn
- These are sweet-tasting, crystalline and soluble in water.
It can be further classified based on:
I. The number of the carbon atom present
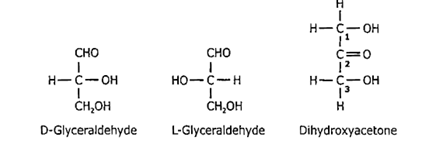
Composed of 3-7 carbon atoms and are classified according to the number of C atoms as trioses (3C), tetroses (4C), pentoses (5C), hexoses (6C) and heptoses (7C).
|
trioses (3C) |
Glyceraldehyde and Dihydroxyacetone |
|
tetroses (4C) – Rare |
Erythrose |
|
pentoses (5C)- Most Common |
Ribose, ribulose, xylulose, and arabinose. |
|
hexoses (6C)- Most Common |
Glucose), fructose, mannose, galactose. |
|
heptoses (7C) |
Sedoheptulose |
II. Presence of aldehyde or ketone group
Suppose a monosaccharide contains an aldehyde group [–CHO]. In that case, it is known as an aldose (reducing centre always lies at Carbon No. 1), and if it contains a keto group [=C=O], it is known as a ketose (reducing centre always lies at Carbon No. 2).
- Optical activity of monosaccharides
All monosaccharides contain one or more chiral carbon atoms (except Dihydroxyacetone), therefore, exist in optically active isomeric forms (enantiomers).
A molecule with one chiral carbon will have two different optical isomers (enantiomers). As the number of chiral carbon increases, the number of possible isomers also increases. Therefore, if a compound has an ‘n’ asymmetric carbon atom, it will have 2n possible stereoisomers.
- Cyclic Forms of Monosaccharides
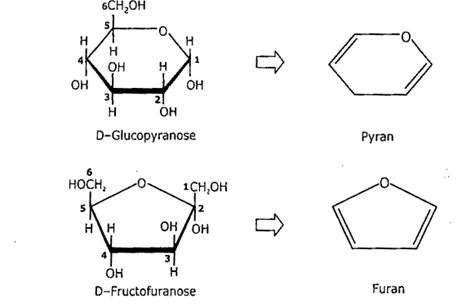
Many monosaccharides, such as glucose, ribose, fructose etc., exist both in open straight-chain and ring form. Sugar with a 5-membered ring is called furanose sugar. Sugar with a 6-membered ring is called pyranose sugar.
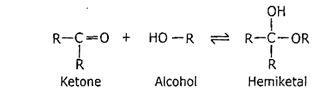
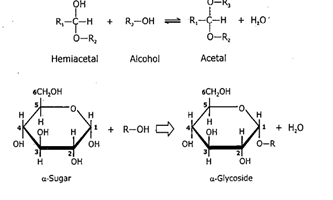
They are formed via internal hemiacetal (aldehyde reacts with alcohol) or hemiketal formation (ketone reacts with alcohol).
Hemiacetal:
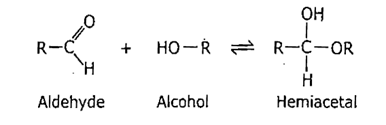
Hemiketal:
The cyclic form of glucose is a six-membered ring; such sugars are called pyranose because they resemble the cyclic form pyran Same in the case of fructose (5-membered rings) called furanose because they resemble furan.
- The hemiacetal or hemiketal bond formation creates a new asymmetric centre at C1 in aldose sugar and C2 in ketose sugar which is now called an anomeric carbon atom.
- The—OH group at the reducing centre (i.e., C No. 1 in aldose sugars and C No. 2 in ketose sugars) is present below the plane of the ring; the sugar is said to be in α-form. And if it is present above the plane of the ring, the sugar is said to be β-form. Thus α-form and β-forms are called Anomers. , The two anomers have different physical and chemical properties.
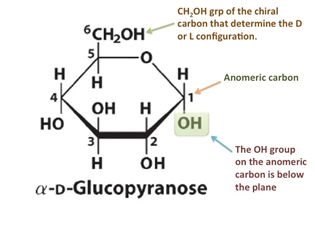
- In an aqueous solution, interconversion of α and β-forms via the open-chain structure gives rise to an equilibrium mixture This phenomenon is called Mutarotation.
EPIMERS
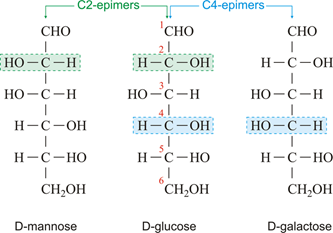
They are called epimers if sugars are closely related but differ only by stereochemistry at a single carbon atom (other than anomeric carbon).
- D- Glucose and D- mannose are epimers because they differ at Carbon 2. Similarly, D- Glucose and D-galactose are epimers because they differ at Carbon 4.
- However, D- Mannose and D- Galactose is not epimers because their configuration differs at more than one carbon.
- Derived Monosaccharides
i. Glycosides: When hemiacetals react with alcohol, they form acetals, and if a hemiacetal of sugar reacts with an alcohol to form acetyl, it is known as a glycoside. It is formed by condensation between the hydroxyl group of anomeric Carbon of monosaccharides, whereas the second compound may or may not be another monosaccharide
Example: Ouabain, which inhibits the enzyme action that pumps NA+ and K+ across the cell membrane
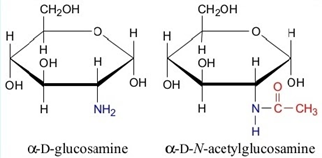
ii. Amino sugar – Hydroxyl group is replaced by an amino or acetylamino group.
Example: Glucosamine, Galactosamine.
iii. Sugar-acid – Aldehyde group at C1 or Hydroxyl group at C6 is oxidized to produce sugar acid. Example: Ascorbic acid, Glucuronic acid (oxidation of glucose), Galacturonic acid (oxidation of galactose)
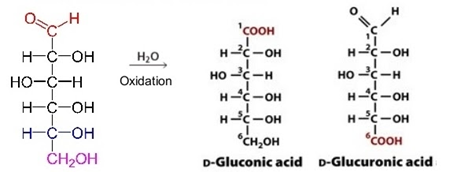
iv. Sugar alcohol – Carbonyl group reduced to the hydroxyl group to form sugar alcohols Example: Glycerol and Mannitol (present in brown algae).
2. OLIGOSACCHARIDES:
Carbohydrates that produce 10 monosaccharide units at max are known as oligosaccharides. They are further classified as disaccharide, trisaccharide, tetrasaccharide and so on
3. Disaccharides:
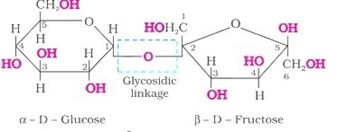
Disaccharides consist of two sugar joined by o-glycosidic bonds. The two monosaccharides are linked by oxide linkage, and such linkage is known as glycosidic linkage. If the reducing groups (aldehyde or ketone) possess bonds, the sugar is non-reducing.
Sucrose is a non-reducing sugar formed as an end product of photosynthesis. In sucrose’s structure, the glycosidic linkage is seen at the C1 terminal of α-D-glucose and C2 of β-D-fructose.
Maltose is prepared by the breakdown of amylase into starch. Maltose is composed of α-D-glucose in which C1 is of glucose and is linked to C4 of another glucose unit.
4. POLYSACCHARIDES:
They are large units of monosaccharides joined together by glycosidic linkages. These molecules are mainly food storage or structural materials. Polysaccharides are broadly classified as homopolysaccharides and heteropolysaccharides.
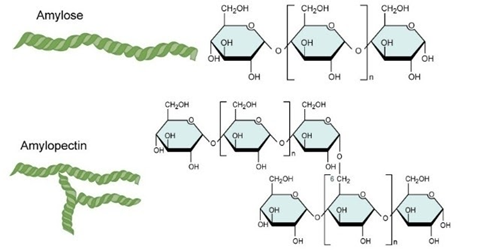
A. Homopolysaccharides are the type of polysaccharides that are formed from only one monosaccharide unit. Starch, Glycogen and Cellulose are examples of homopolysaccharides. Starch is used as the glucose storage form of carbohydrates in plants. It contains a mixture of amylase and amylopectin. Amylose is linear unbranched polymer, whereas amylopectin is a branched polymer of glucose.
Glycogen is used as the glucose storage for carbohydrates in animals, mostly found in the liver and muscle. It is also made up of glycogen and glucose. It is composed of α-1,4-glycosidic bonds with an α-1,6 bond present, which is seen at every tenth monomeric unit.

Structure of Glycogen
Cellulose is another example of a polymer of glucose and is a linear, branched homopolysaccharide of D- glucose units of beta-1, 4-glycosidic linkage. It strengthens the cell wall. Some common forms of cellulose known to us are paper and wood.
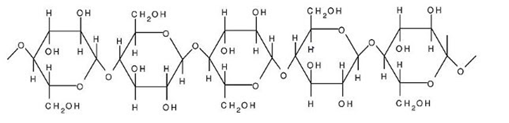
Structure of cellulose
B. Heteropolysaccharides; type of polysaccharides that are made up of different monosaccharide units. Peptidoglycan and hyaluronic acid are some examples of heteropolysaccharides. Peptidoglycan or murein; is responsible for forming the bacterial cell’s cell wall and chitin is responsible for forming the exoskeleton of arthropods, whereas Hyaluronic acid can work as a lubricant in joint’s synovial fluids.
Download PDF for CSIR-NET Short Notes on CARBOHYDRATES – Click Here
Also Read :
Short Notes on Carbohydrates and It’s Three Main Categories
Short Notes on Mechanisms of Enzyme and Enzymatic Regulation!
~ Keep Upvote this article and share it among your friends.
→ If you have any questions feel free to ask in the comments section below.
Join us on our Telegram Channel for daily updates.
Subscribe to our Youtube Channel also for daily Live Classes for CSIR NET 2021.
BYJU’S Exam Prep
Download the BYJU’S Exam Prep App Now.
The Most Comprehensive Exam Prep App.
#DreamStriveSucceed
App Link: https://bit.ly/3sxBCsm


